PRP therapy has been getting a lot of attention in Canada as of late. Just like in the U.S., Canadian regulators are attempting to clamp down on regenerative medicine clinics offering PRP and stem cell therapies. To that end, Health Canada recently clarified its position that PRP therapy is a form of drug therapy.
You can look at Health Canada’s position as being both good and bad. In a perfect world, regulators would take no position at all. But given that this world is not perfect, regenerative medicine practitioners and patients alike have to work within the confines of the regulatory environment.
Note that there has long been disagreement in Canada over whether PRP therapy is a drug therapy or one in the same league as stem cell therapy. Health Canada felt it necessary to clarify its position so as to address that disagreement. They have succeeded in that much. Yet now there is disagreement over whether PRP therapy should be regulated at all.
The Good
Let’s talk about the good aspects of Health Canada’s position first. Understand that drug therapies in Canada are regulated less strictly than more advanced treatments. In terms of PRP therapy, classifying it as a drug therapy means that clinicians and researchers don’t have to adhere to some of the stricter stem cell regulations.
Another benefit is that proponents of PRP therapy do not have to prove efficacy. That is a pretty significant burden to be freed from. As long as PRP therapy is administered according to current regulatory standards, it is considered safe. That’s all Health Canada is worried about at this point.
The end result is that more clinics and doctors might start offering the therapy. Now that they know where they stand from a regulatory standpoint, they know what they can and cannot do with PRP. Perhaps this is just the kick in the pants Canada’s regenerative medicine industry needs.
The Bad
While we can look at Health Canada’s position in a positive light, there is always some bad that comes with the good. The bad here points back to the central question of whether government should be involved in regulating PRP therapy. And if so, should regulation be kept to a minimum?
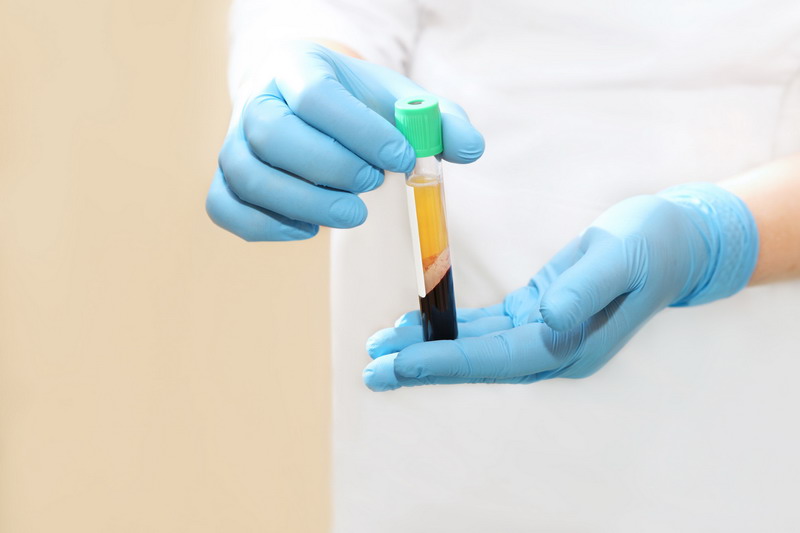
The problem with classifying PRP as a drug is pretty simple: PRP is not a drug. According to Utah-based Apex Biologix, platelet-rich plasma is derived by drawing blood from a patient then spinning the blood in a centrifuge to isolate plasma and platelets. That’s it. The resulting material extracted from the centrifuge is the same material drawn from the patient but with unwanted cells removed.
If it’s okay to classify PRP as a drug, then why not also classify the skin used for skin grafts as a drug? Why not just classify all human tissue as a drug? That is essentially what Health Canada is saying by taking the position that PRP therapy is a drug therapy.
If you do not understand why this is a bad thing, just step back and consider the ultimate purpose. Health Canada takes its position only to assert its authority to regulate PRP therapy in whatever way it sees fit. Whether they regulate minimally or take things to the extreme is not the point here. The point is that they have classified PRP is a drug therapy so that they can control it.
This is central to the question of whether or not PRP therapy should be regulated at all. It is a question we are a long way from answering both in Canada and the U.S.





More Stories
Orange County CA Sober Living for Long-Term Recovery Success
How Swimming Supports Children With Sensory Sensitivities
Gain Peace of Mind Through Reliable Medicare Advantage Plans 2026